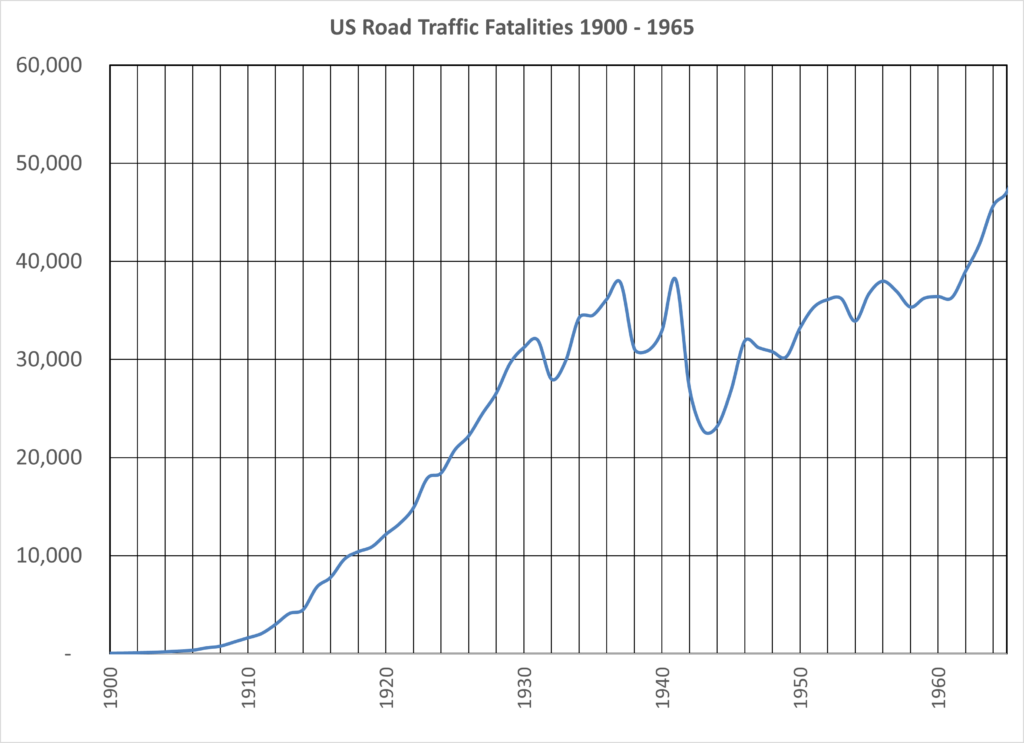
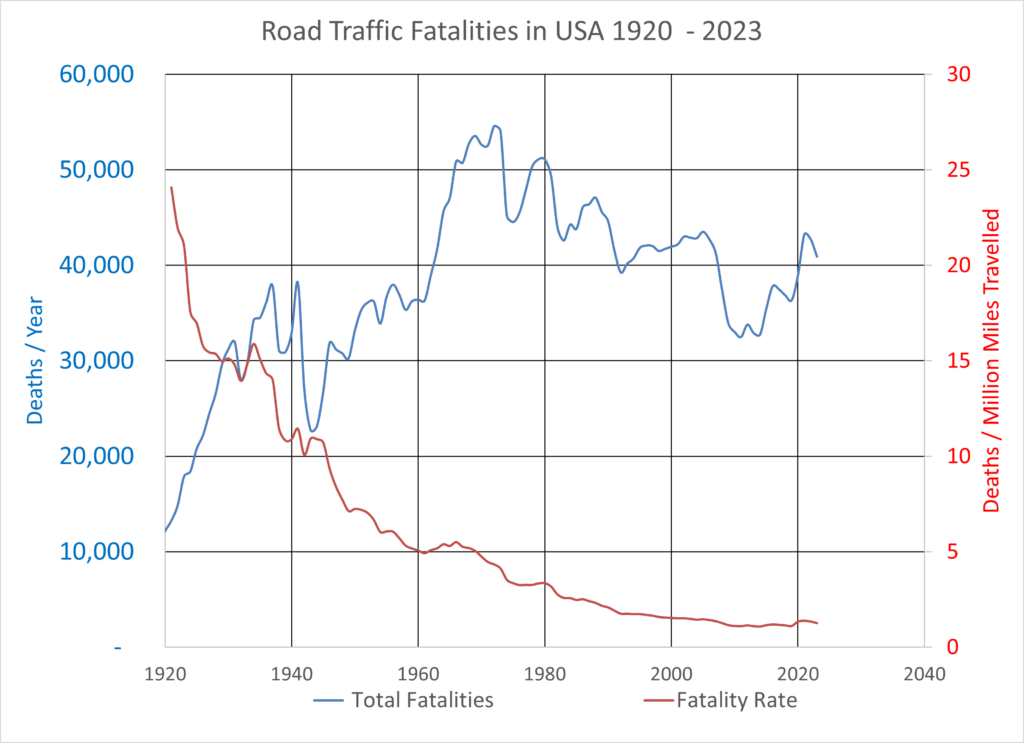
How should we balance the rights and obligations of consumers, – who may be harmed by dangerous or unsafe products, – and the companies who produce and distribute the products we need?
In the UK, there are three ways that consumers who are harmed by dangerous and unsafe products can claim compensation from the manufacturer or distributor.
They may be able to make claim for:-
- Breach of Contract, if they can show the the supplier ‘did not do what they promised’, because the product is ‘not fit for purpose’ or does not comply with the agreed specification. Claims for breach of contract will depend on the terms and conditions of the contract – or sale agreement – and any warranties that were given at that time, but can only be made by people who are ‘parties to the contract’.
- Negligence, if they can show that they were owed a ‘Duty of Care’, and the producer failed to take reasonable care to prevent the ‘foreseeable risk of harm’ that caused their actual loss or injury.
- Product Liability, if they can show that the product was defective, because it didn’t provide the level of safety they were entitled to expect, and that the defect caused their loss or injury. These claims are based on Part 1 of the Consumer Protect Act 1987, which implemented in UK Law the EU’s original product liability directive, including the principle of the Producer’s Strict Liability (without fault) for defective or unsafe products.
So, consumers may plead their case based on any combination these 3 arguments, provided they can find enough supporting evidence. But commercial losses and damage to commercial property are excluded from product liability claims based on Part 1 of the Consumer Protection Act 1987 – because it is only intended to protect the rights of natural born people – not corporations.
To supply safe products, producers need to comply with the UK’s General Product Safety Regulations – which implemented the EU’s General Product Safety Directive 87/357 and any other product specific rules and regulations – including the use of UKCA or CE marking on regulated products.
Now, following BREXIT, the Government is proposing a major overhaul of the UK’s product liability law, redefining the rights and obligations of consumers and economic operators who supply and distribute the goods and service we need. This will address the following problems identified by the UK government:-
- Our increasing dependence on software and digital services, which may control the function and performance of more traditional ‘physical products’, introducing new or additional risks that were not considered when the current legislations was created.
- The sale and distribution of products through on-line channels and market places, where the ‘producer’ may not be present in the UK.
- The increasing use of recycled products and components to reduce waste.
- Our departure from the European Single Market and independence from the EU, who have recently updated EU Product Liability Law to address some of the same concerns, with a new General Product Safety Regulation (EU) 2023/988 and Product Liability Directive (EU) 2024/2853
The UK Government has already announced that it will repeal Part 1 of Consumer Protection Act 1987, and replace it with a New Regulation – without defining it’s scope or content of any new legislation is now under review.
So, the Law Commission is reviewing the UK’s Product Liability legislation, starting with a public consultation to help define the scope of their enquiry and any subsequent changes.
So, they are inviting you to answer the following questions –
Question 1: Do you think that the Product Liability regime provided for by Part 1 of the Consumer Protection Act 1987 works well? Please explain the basis for your view i.e. why you do or do not.
Question 2: If you consider that the Product Liability regime provided for by Part 1 of the Consumer Protection Act 1987 needs to be reformed, please set out how you think it should be reformed. Please also provide the rationale for your proposals.
Question 3: Are you concerned about potential reforms to the Product Liability regime provided for by Part 1 of the Consumer Protection Act 1987? If you are concerned, please set out what potential reforms concern you with an explanation why.
But, we face a ideological choice – should UK law and regulation be closely aligned with the EU reflecting their new Product Liability Directive and updated General Product Safety Regulation? or should we diverge from the ‘European Model’ whilst searching for the intangible benefits of BREXIT?
Repealing Part 1 of the Consumer Protection Act 1987, may be than a symbolic gesture. This legislation introduced, into UK Law, the concept of ‘Strict Liability’ for the supply of dangerous and unsafe products, holding manufacturers and their component suppliers jointly liable for any harm caused by the product lack of safety.
So what should replace it?
Update – 24 December 2024
The Law Commission have now published the Terms of Reference for it’s review of the UK’s Product Liability legislation. Although the issues raised were previously addressed by the EU when formulating the New Product Liability (2024/2853/EU), we should not assume the UK will reach same conclusions or choose alignment with the EU as it preferred or default strategy.
Learn more…
Stunell Technology deliver product liability training for engineers and engineering managers responsible for product development, manufacturing operations or aftermarket support.
To discuss your team’s training requirements, please contact Phil Stunell or book an on-line appointment